
Steven Lee/Whitehead Institute
A Fruitful Pursuit: Plant Biology at Whitehead Institute
Plants have been a potent source of medical and scientific discovery for centuries. In the 19th century, Gregor Mendel’s famous experiments on trait inheritance in pea plants, later expanded on by other botanists, laid the groundwork for the discovery of genes. Much later, the first transposon, or jumping gene, was also discovered in plants.
Plant research continues to bear rich fruit today. Advances in biochemistry and genomics are identifying plant products that lead to new pharmaceuticals, improvements to crop yield and food security, and a better understanding of many biological processes—from gene regulation to protein folding—providing insights that may extend to humans and other species.
Whitehead Institute has appreciated the importance of plants as research subjects since its inception, with its top-floor greenhouse a core facility of the Institute, and its researchers have made valuable contributions to the field of plant biology. Read about some of that research below and to learn more, follow the links to recent news pieces, podcasts, and videos.
Making a model
 |
| Arabidopsis thaliana in Whitehead Institute's greenhouse Image: Conor Gearin/Whitehead Institute |
In the 1980s and 1990s, Whitehead Institute Founding Member and former Director Gerald Fink helped to establish Arabidopsis thaliana, commonly known as mouse-ear cress, as a model organism — a species that is a good research tool for investigating biological processes in the lab. Arabidopsis has become a go-to organism for plant research, thanks to advantages such as its small genome size, quick life cycle, and ease of cultivation.
Fink used Arabidopsis to identify a mechanism of hormone signaling, determine how the plants use gravity to orient their growth, and engineer an Arabidopsis plant that can survive drought and saltwater conditions. Whitehead Member David Bartel and former Whitehead Member and Director Susan Lindquist have also used Arabidopsis to aid in their respective research pursuits. After the discovery of microRNAs, very small RNA molecules that can help regulate genetic expression, Bartel began investigating the molecules’ prevalence and function. These questions at first proved easiest to answer in plants. Meanwhile Lindquist used Arabidopsis to, among other things, examine the role of heat-shock protein 90 in mediating expression of genetic variation.
 |
| Postdoc Benjamin P. Williams Credit: Conor Gearin/Whitehead Institute |
However, plant research at Whitehead really blossomed with the arrival of Mary Gehring as a Whitehead Member in 2010, followed in 2013 by Jing-Ke Weng. With the addition of their research programs, Whitehead Institute gained a cohort of researchers dedicated to plant science. Gehring and Weng’s research has made impactful discoveries revealing how plant genes are regulated, illuminating aspects of their evolution, and providing leads for potential plant-based medical and agrochemical products.
Beyond the gene
 |
| Whitehead Institute Member Mary Gehring Credit: Gretchen Ertl/Whitehead Institute |
Gehring uses Arabidopsis to investigate epigenetics, or how gene expression can be altered without altering the DNA sequence of the gene. Much of Gehring’s work focuses on DNA methylation, an epigenetic mechanism in which small chemical tags attach on or near genes and by so doing alter their expression, turning them on or off. In plants, DNA methylation can be inherited much like genes, meaning that epigenetic changes can shape future generations. This heritability has intrigued Gehring, whose lab is pursuing how these mechanisms work. Clues came in 2017, when Gehring and postdoctoral researcher Ben P. Williams published a paper on the gene ROS1, which appears to function as a rheostat maintaining a steady, heritable level of methylation on genes.
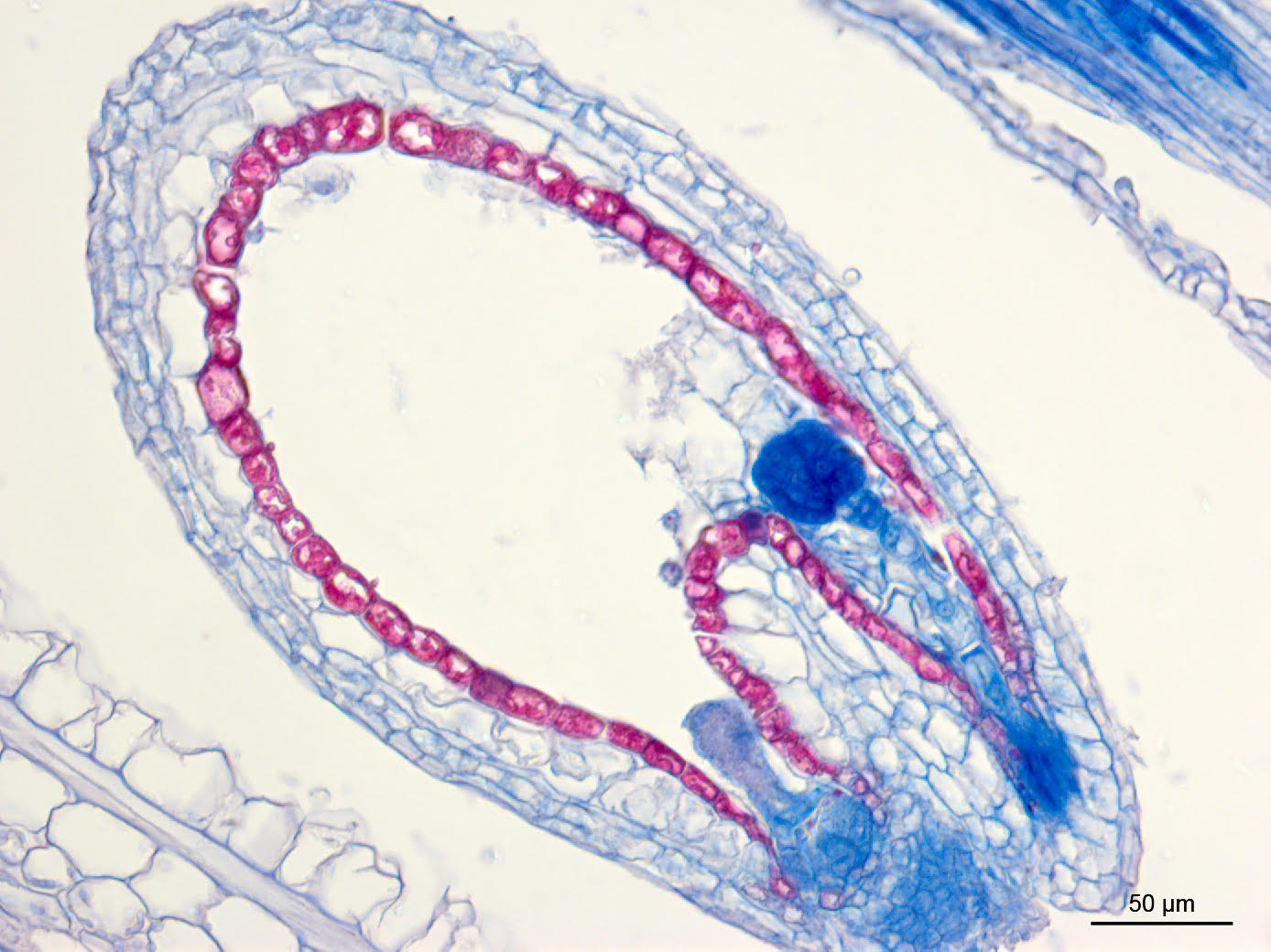 |
| Microscope image of atypical development in misimprinted A. thaliana seed Credit: Katherine Novitzky/Whitehead Institute |
Gehring also investigates imprinting, in which a gene’s expression can be biased to favor the copy of a gene inherited from the plant’s male or female parent. Upon fertilization, plants receive copies of each gene from their mother and father. In imprinted genes, the copies donated by each parent have different levels of methylation such that one parent’s copy is silenced and only the other parent’s copy is expressed. Recent work from Gehring and former Gehring graduate student Robert Erdmann shows that this process is sometimes regulated by small RNAs. The researchers found that if small RNA production is disabled, the balance of expression between genes from each parent is thrown off.
Another study by Gehring and former lab members Daniela Pignatta and Katherine Novitzky shows that imprinting can lead to differences in the characteristic traits between plant strains, including the timing of seed development and seed size.
To hear more about Gehring’s work in her own words, listen to her on Whitehead’s AudioHelicase podcast.
Nature’s medicine
Since plants cannot move, they have instead developed an arsenal of natural products to compensate, helping them repel predators, attract pollinators, and interact with their environments. Many of these chemicals have medicinal or other beneficial properties, and countless plants have been used for thousands of years in traditional global medicine. Weng investigates these plants to discover potential therapeutics and other possible products. However, harnessing their potential at scale requires overcoming challenges in identifying, producing, and harvesting the chemicals in an environmentally-sustainable way.
 |
| Whitehead Institute Member Jing-Ke Weng Credit: Gretchen Ertl/Whitehead Institute |
The natural product salidroside, produced by the golden root (Rhodiola rosea) plant, has been used for thousands of years to treat depression and fatigue. Weng and postdoctoral researcher Michael Torrens-Spence determined how golden root produces salidroside, tracing back the steps to find the collection of genes responsible. They then transferred these genes into both tobacco plants and baker’s yeast, model systems that grow quickly and can mass-produce the chemical. Weng hopes that this production model will be widely adopted, as it not only allows for increased production at the levels necessary for pharmaceutical testing and development, but it also could protect endangered plant species that produce valuable chemicals from being overharvested.
 |
| Goji berries Credit: Lycium barbarum by Andreas Rockstein, CC BY-SA 2.0 |
Weng, postdoctoral researcher Fu-Shuang Li, and collaborators have also elucidated the chemical structures of some of the more complex and elusive plant metabolites, such as sporopollenin, a biopolymer responsible for the tough, protective outer shell of plant spores and pollen grains. This research opens up avenues to engineer new nature-inspired materials, such as anti-fouling paint for ship hulls and inert coatings for medical implants.
Certain plants, such as soybeans and canola, have been genetically engineered to carry an enzyme called BAR in order to make them immune to a common herbicide. This allows farmers to spray their fields without damaging the crop. However, Weng and postdoctoral researcher Bastien Christ found that BAR caused unintended side effects in the plants carrying it. They re-engineered the enzyme to make its effects more specific and potentially have fewer unintended effects.Weng’s lab has used similar approaches to identify the genes behind other metabolites.
Recently Weng and postdoctoral researcher Roland Kersten identified various lyciumins, a natural product found in goji berries and other plants that might be useful for managing blood pressure.
To learn more about Weng’s research, watch this multimedia piece featuring his work.
Contact
Communications and Public Affairs
Phone: 617-452-4630
Email: newsroom@wi.mit.edu


