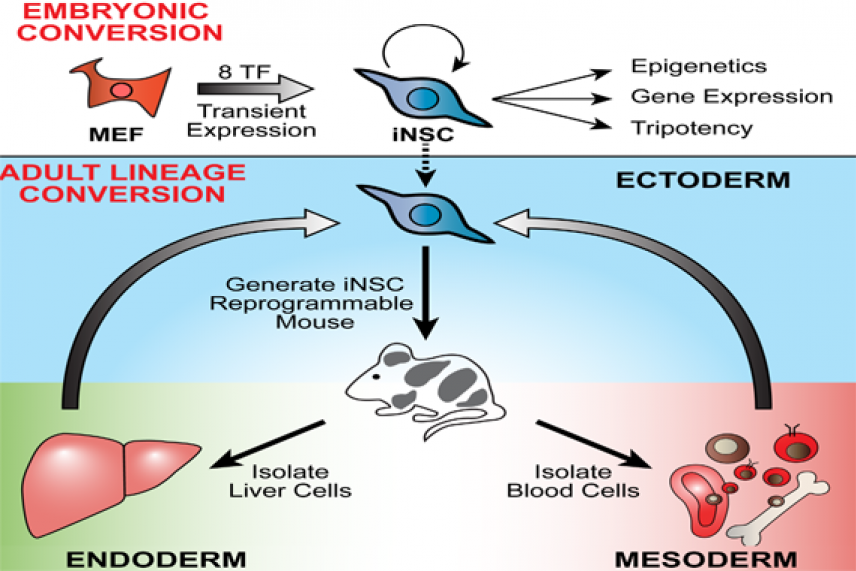
This graphic depicts how scientist in Whitehead's Jaenisch lab generated induced neural stem cells (iNSCs) that are transcriptionally, epigenetically, and functionally similar to primary NSCs by transiently overexpressing transcription factors in mouse embryonic fibroblasts (MEFs). The scientists then demonstrated lineage conversion by inducing tripotent iNSCs from a variety of adult cell types, including liver cells and B-cells with genetic rearrangements.
Courtesy of Cell Press
Direct generation of neural stem cells could enable transplantation therapy
CAMBRIDGE, Mass. – Induced neural stem cells (iNSCs) created from adult cells hold promise for therapeutic transplantation, but their potential in this capacity has been limited by failed efforts to maintain such cells in the desirable multi-potent NSC state without continuous expression of the transcription factors used initially to reprogram them.
Now, Whitehead Institute scientists have created iNSCs that remain in the multi-potent state without ongoing expression of reprogramming factors. This allows the iNSCs to divide repeatedly to generate cells in quantities sufficient for therapy.
“Therapeutically, it’s important to make neural stem cells because they can self-renew and make lots of cells,” says Whitehead Institute Founding Member Rudolf Jaenisch, who is also a professor of biology at MIT. “If you just make mature neurons, which has been done by others, you never get enough cells.”
To make iNSCs via direct lineage conversion researchers use viruses to insert a cocktail of transcription factors into the genome of mouse adult skin cells. A drug triggers these transcription factors to turn on genes active in neural stem cells. This direct conversion, known as transdifferentiation, bypasses the step of pushing the cells first through an embryonic stem-cell-like state.
In previous research, iNSCs remained addicted to the drug and reprogramming transcription factors; if either the drug or the factors were removed, the cells reverted to skin cells or spontaneously differentiated.
“If the reprogramming factors are still active, it’s horrible for the cells,” says John Cassady, a scientist in Jaenisch’s lab. “The cells would be unable to differentiate and the resulting cells would not be therapeutically useful.”
In a paper published online this week in the current issue of the journal Stem Cell Reports, Cassady and other Whitehead scientists describe how they preserve the cells’ properties without keeping the reprogramming factors active. First, the cells were grown in a special medium that selects for neural stem cells. Then, the drug is removed. Instead of spontaneously differentiating, the iNSCs remain in a multi-potent state that can differentiate into neurons and glia cells on cue. Cassady also refined the reprogramming cocktail to contain eight transcription factors, which produces iNSCs that are transcriptionally and epigenetically similar to mouse neural stem cells.
Cassady notes that a random sample of skin cells can contain neural crest cells, which may more easily make the transition to iNSCs. To eliminate the possibility that his method might actually rely on cells having this sort of “head start”, Cassady converted fully mature immune system cells called B-lymphocytes, which have a very specific genetic marker, to iNSCs. The resulting cells had the profile of their new identity as iNSCs, yet retained their B-lymphocyte genetic marker, showing that Cassady’s method could indeed convert non-neural cells to iNSCs.
Although promising, all of the work to date has been conducted in mouse cells. According to Cassady, researchers should next test this protocol in human cells to see if it can successfully produce the cell populations necessary for therapeutic use.
This work is supported by Howard Hughes Medical Institute (HHMI) and the National Institutes of Health (NIH; grants HD 045022, R37CA084198, and HG002668).
* * *
Rudolf Jaenisch's primary affiliation is with Whitehead Institute for Biomedical Research, where his laboratory is located and all his research is conducted. He is also a professor of biology at Massachusetts Institute of Technology.
* * *
Citation:
Topics
Contact
Communications and Public Affairs
Phone: 617-452-4630
Email: newsroom@wi.mit.edu


