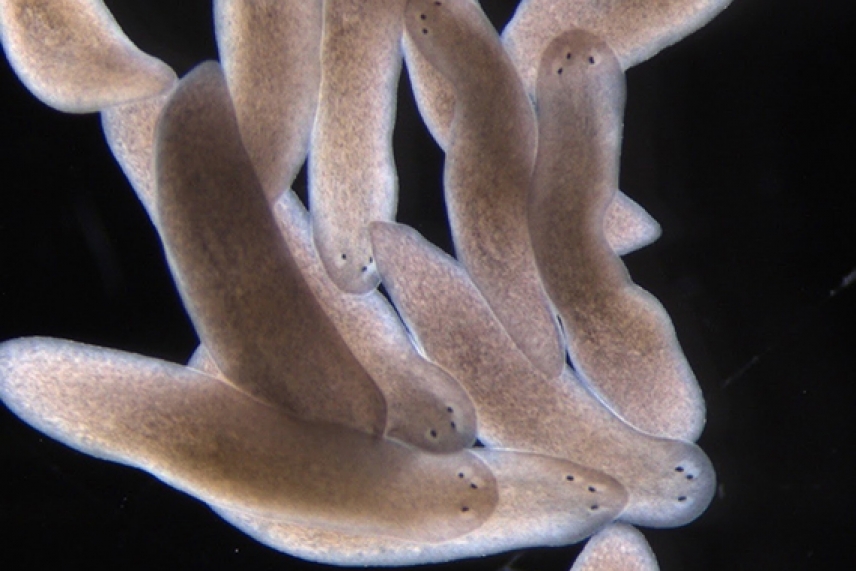
Three-eye planarians generated by a simple surgical trick revealing self-organizing dynamics that occur during regeneration.
Kutay Deniz Atabay/Whitehead Institute
New tools for an old can of worms
This lab exercise is simple—although not necessarily for the squeamish.
Take one live planarian flatworm. Chill it nicely in an iced Petri dish. Using a tiny microsurgical blade, cut the centimeter-long beast into as many pieces as you can stand, and plop them in a plastic bowl. Store it in the dark.
After a week, you'll see fully formed planarians swimming around in your bowl.
For many decades, scientists have been carrying out versions of this experiment and banging their heads against the wall trying to understand the result. It turns out that you can cut off a planarian slice as small as 1/279th of the animal and have it turn into a complete adult. (That finding, by the way, comes from Thomas Hunt Morgan, who gave up on planarian regeneration studies in frustration and turned to his pioneering work in the genetics of fruit flies.)
For many centuries, inquiring minds have puzzled over the capabilities of certain worms, amphibians, fish and other animals to regrow limbs and other body parts. Serious scientific inquiry goes all the way back to 1740, when Abraham Trembley experimented with hydra. But while researchers carefully documented, for example, exactly what happens if you cut planarians into quarters, the underlying mechanisms remained entirely mysterious. And while developmental biology has exploded in recent decades, the field of regeneration biology remains rather small.
"Regeneration is one of the great mysteries of biology that has puzzled developmental biologists for well over a century," says Whitehead Associate Member Peter Reddien. But that's changing quickly as researchers bring the powerhouses of modern biological analyses to studying these processes-with the hope that a better understanding of regeneration may eventually find medical applications.
How the worm turns over its cells
Planarians such as Schmidtea mediterranea live in freshwater streams and ponds, eating insects and avoiding light, which they spot with the simple photodetectors that give them a cross-eyed look. Their digestive and nervous systems appear rudimentary.
But how they reproduce is not rudimentary at all. Some strains reproduce as cross-fertilizing hermaphrodites (both worms containing both sperm and eggs and simultaneously fertilizing each other). Other strains reproduce by dividing in half, with both head and tail forming a new animal much like the amputated chunks.
New planarian tissues and organs are created by neoblasts-adult stem cells that share certain characteristics with embryonic stem cells and can differentiate into essentially all the cells in adult animals. This process occurs in both chopped-up and normal worms. Just how it works is still almost completely unknown.
Biology's major model organisms aren't much help either, declares Alejandro S‡nchez Alvarado, Howard Hughes Medical Institute investigator and professor of neurobiology and anatomy at the University of Utah. (Reddien worked as a postdoc in S‡nchez Alvarado's lab before arriving at Whitehead this year.)
The C. elegans worm and the Drosophila fruit fly, "the warhorses of developmental biology," flunk out on this test. "Pull a wing off of a Drosophila, and it won't grow back," says S‡nchez Alvarado. "Take a C. elegans and slice it in half, and it will die."
S‡nchez Alvarado has been working since the late 1990s to turn S. mediterranea into a model organism. He chose the worm for its regeneration capabilities, developmental plasticity, ability to reproduce both sexually and asexually, and its unusual stem cells. It didn't hurt that S. mediterranea is relatively easy to work with in the lab, and that it later turned out to perform functions with relatively few genes per function. He and his coworkers have been steadily accumulating knowledge through new laboratory tools.
RNAi and the regeneration gap
Most dramatically, they've employed the use of RNA interference (RNAi), a technique that can knock out the function of individual genes.
Reddien led the first high-throughput RNAi screen of planarian genes during regeneration, with results published this May in Developmental Cell. The researchers painstakingly silenced 1,065 genes one at a time, and found that 240 of these genes, when silenced, produced defects in the worms.
No fewer than 204 had corresponding genes in other species. "There's a large degree of conservation between the genes that are affecting regeneration efficiency in planarians and genes in C. elegans, Drosophila and humans," says S‡nchez Alvarado. That's intriguing because highly diverse organisms often develop via very similar molecular pathways. "Neurons and muscle cells, say, all are made much the same way by hydras and humans," he points out.
While 145 of the silenced genes were essential for regeneration and homeostasis (normal tissue loss and replacement), other genes were required for one or the other but not both. This suggests separate molecular pathways for homeostasis and regeneration-an encouraging sign for regeneration studies.
The team also found a wealth of leads for further research, including a novel gene apparently involved in wound healing.
At Whitehead, Reddien is plunging ahead with additional RNAi screening, which "works incredibly well for planaria," he says. He and his colleagues have found they can express double-stranded RNAs in bacteria, and introduce the bacteria into the planarian's liver-and-agar lunches. The double-stranded RNA spreads through all cell types and shuts off the targeted gene.
The S. mediterranea genome is being sequenced at the Genome Sequencing Center at Washington University in St. Louis. S‡nchez Alvarado, Reddien and Philip Newmark wrote the white paper that brought funding from the National Human Genome Research Institute.
For now, though, "we are still amazingly ignorant about the cell biology of what's happening in regeneration," cautions Newmark, an assistant professor of cell and structural biology at the University of Illinois in Urbana.
"Regeneration is in the same state as developmental biology was at the start of the 20th century," as S‡nchez Alvarado puts it. With scientists just starting to unveil the molecular frameworks for cellular processes, "we have no clear pictures of what the stem cells are doing throughout the day," he says.
Planarian puzzles
"Planarians have really challenged the way I thought about biology," says Whitehead's Peter Reddien. "They're incredibly fascinating animals."
"The animals really defy what we learned in graduate school," agrees Alejandro S‡nchez Alvarado of the University of Utah. "You watch in the microscope and it just boggles the mind."
Amazing worm tricks:
•Reproducing asexually by splitting in half, "they can maintain the immortality of the species without ever going through a germ line," Reddien says. "Adult tissues can therefore live essentially forever."
•Sexual strains don't generate the cells that are dedicated to making sperm and eggs as well as their sexual organs until they reach a certain size.
•"If you starve these animals, they'll shrink," says Reddien. "We call it de-growth, because it's exactly the opposite of growth. It's not just a change in cell mass or cell size, it's a change in cell number. They're eliminating cells from intact organ systems, shrinking the tissues-their brain, their intestine, their skin-while maintaining the form and function. And then if you feed animals that have shrunk, they'll grow again."
This article first appeared in the Fall 2005 issue of Paradigm magazine.
Topics
Contact
Communications and Public Affairs
Phone: 617-452-4630
Email: newsroom@wi.mit.edu


