Figuring out the heads or tails decision in regeneration

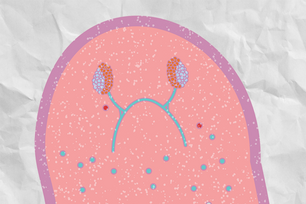
Planarian with wntP-1 concentration at wound site
PNAS (Petersen and Reddien) Published online before print on September 14th, 2009
CAMBRIDGE, Mass. – Amputations trigger a molecular response that determines if a head or tail will be regrown in planaria, a flatworm commonly studied for its regenerative capabilities. Until now, no molecular connection between wounding and the decision to regenerate either a head or tail in planaria had been identified.
Whitehead Institute scientists report this finding in the September 15-28 issue of PNAS Early Edition.
Regeneration is the regrowth of part of an organism’s body after it has been damaged or removed. Although animals such as planaria and starfish can regrow virtually any part of their bodies, humans have restricted regenerative capabilities. A better understanding of regeneration, including how the body decides which organs or tissues should be regrown after an injury, may eventually benefit patients with traumatic injuries, such as amputations or nerve damage.
To study how planaria regrow their bodies from a sliver of tissue, Christian Petersen, a postdoctoral fellow in Whitehead Member Peter Reddien’s lab, looked at two of the planaria’s genes, wntP-1 and wntP-2. These genes act to control cell to cell communication in a process called the Wnt signaling pathway, which is involved in many biological events, including development and growth. Petersen and Reddien had theorized that at least some portion of the Wnt pathway plays a role in regeneration.
In earlier work with planaria, Petersen and Reddien showed that a Wnt-related gene, called Smed-beta-catenin-1 (beta catenin), is necessary for planaria to regenerate a tail instead of a head after tail amputation. This ability to grow a head when the head is cut off and grow a tail when the tail is cut off is known as “regeneration polarity”. In this experiment, Petersen cut the head, tail or both the head and tail off of planaria and observed where wntP-1 gene expression occurred.
Because previous experiments linked the Wnt-related gene beta catenin to proper tail regrowth, Petersen and Reddien expected that only the worms with amputated tails would express the wntP-1 gene. However, shortly after wounding the worms, a number of cells near both the head-facing and tail-facing wound sites expressed the wntP-1 gene.
“That was quite surprising, and it suggested that maybe wntP-1 is expressed at any wound,” says Petersen. He then created a series of other wounds that did not remove an entire head or tail, and saw the same result: wntP-1 was expressed at all of the wound sites. After Petersen blocked the gene, only heads regrew in the place of tails, forming two-headed planaria. Investigating further, Petersen and Reddien found that in planaria with their tails cut off, the wntP-1 gene triggers beta-catenin, which activates the wntP-2 gene. If any of these steps is blocked, a head regrows instead of a tail.
Although Petersen and Reddien have figured out part of the regeneration process in planaria, the finding is only the beginning.
“We really want to understand the mechanism for how the polarity decision is made during regeneration – how is it specified that a the wound is facing the old head or facing the old tail?” asks Reddien, who is also an assistant professor of biology at Massachusetts Institute of Technology and a Howard Hughes Medical Institute (HHMI) early career scientist. “We think this question is very fundamental in understanding the determination of tissue identity. And this paper is an important step in understanding that mechanism.”
This research was funded by the National Institutes of Health (NIH), the American Cancer Society, the Rita Allen Foundation, the Searle Scholars Program, the Smith Family Foundation, and the W. M. Keck Foundation.
* * *
Peter Reddien’s primary affiliation is with Whitehead Institute for Biomedical Research, where his laboratory is located and all his research is conducted. He is also a Howard Hughes Medical Institute Early Career Scientist and an assistant professor of biology at Massachusetts Institute of Technology.
* * *
Citation:
Petersen, C. P., & Reddien, P. W. (2009). A wound-induced Wnt expression program controls planarian regeneration polarity. Proceedings of the National Academy of Sciences, 106(40), 17061-17066.
Contact
Communications and Public Affairs
Phone: 617-452-4630
Email: newsroom@wi.mit.edu